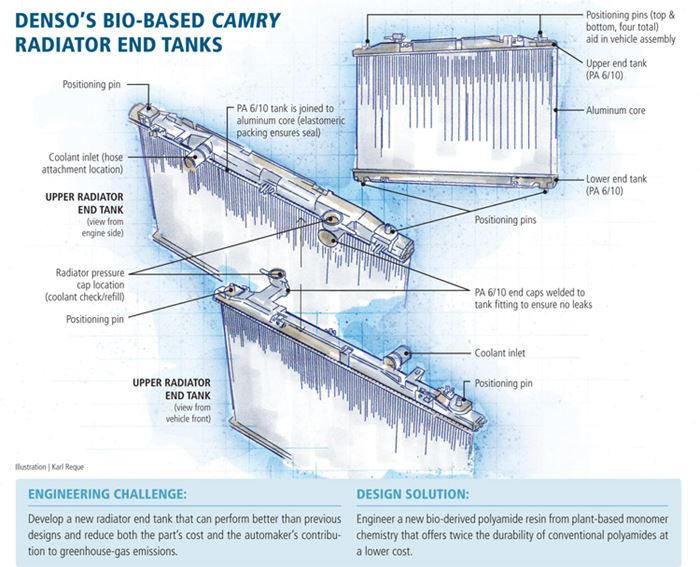
Cleaner and greener: Bio-based end tanks
Plant-based polyamides break high-cost/low-performance paradigm to meet the demands of a challenging Toyota underhood application.
Engineering Challenge:
Develop a new radiator end tank that can perform better than previous designs and reduce both the part’s cost and the automaker’s contribution to greenhouse-gas emissions.
Design Solution:
Engineer a new bio-derived polyamide resin from plant-based monomer chemistry that offers twice the durability of conventional polyamides at a lower cost.
Automakers worldwide are scrambling to improve vehicle energy efficiency and reduce or offset greenhouse-gas emissions. Composites are a proven means to trim part weight, a key strategy in the quest for fuel savings, and they also can reduce greenhouse emissions if bio-based monomers are used to formulate the resin matrix. The use of annually renewable plant-based chemistry helps greatly with carbon mitigation. Because growing plants absorb carbon dioxide (CO2) — one of the most common greenhouse gases — during their lifecycle, plants harvested for use in bio-polymers or for natural fiber reinforcements help offset the emissions released both during production of the part and the vehicles on which the part will be installed. If the usage is significant enough, some OEMs claim, plant-based matrices and reinforcements will offset a portion of the emissions released during the vehicle’s life. If the U.S. and other big greenhouse gas emitters begin strict enforcement of carbon caps, then the practice of trading emissions credits, earned through renewable energy generation and the use of products like bio-polymers and natural fiber reinforcements, could move beyond “green buzz” to something with serious economic value.
Bio-based history
Although Henry Ford is credited with the first use of bio-based polymers in auto parts on his Soybean Car demonstrator in 1941, their use in commercial passenger cars dates from the 1990s. Much modern usage was pioneered by Toyota Motor Co. (Aichi, Japan) and, later, Ford Motor Co. (Dearborn, Mich.). Initially, the focus was polylactic acid (PLA), a biodegradable thermoplastic polyester similar to polyethylene terephthalate (PET) but with lower thermal performance. That meant that applications were confined to small parts, with primarily decorative functions, located below the beltline (below seatbelt level, where sunlight is less likely to heat the plastic). Early on, biopolymers earned a reputation for being more expensive and less durable than conventional polymers, which slowed market acceptance. As with biofuels, much of the early work involved feedstocks from food crops (e.g., corn or soy) that otherwise could feed humans or livestock. This drew criticism for its potential to drive up food prices in the developing world, thereby negating the social and environmental benefits gained by offsetting petroleum products.
However, in the past decade, more applications of a broader range of chemistries have been introduced as resin suppliers have identified plants that can be coaxed into producing chemicals that are useful in formulating resins capable of higher performance. These have piqued the interest of automakers and systems suppliers.
One of the most innovative and challenging bio-composites applications to date is found in radiator end tanks that debuted on the 2010 model year (MY) Toyota Camry sedan. The application was jointly developed by systems supplier, toolmaker and molder DENSO Corp. (Kariya, Aichi, Japan) and resin supplier DuPont Automotive (Troy, Mich.). The parts are injection molded from a 30 percent short glass-reinforced blend of polyamide (PA, also called nylon) 6/10 resin formulated with 40-percent bio-based monomers obtained from castor beans, a nonfood crop with medicinal, insecticidal and ornamental uses that also was used as a motor lubricant during World War I. The Camry’s radiator end tanks are a compelling illustration of the fact that bio-plastics can meet demanding mechanical, thermal and chemical requirements.
Biopolymers in the hot seat
When DENSO and DuPont began their four-year collaboration (circa 2005), the goal was to find a plant-based material that could be used in radiator end tanks. The tanks were a difficult application for conventional plastics and typically required heat-stabilized, hydrolysis-resistant grades of PA 6/6, a blend of 6/6 with 6/12, or 6/10. Mounted directly onto the metal radiator core, the tanks form part of the radiator’s circulatory loop. Tank materials are in constant contact with often hot ethylene or propylene glycol — chemicals not known for kindness to polymer backbones, especially that of PA. They also are exposed to salt spray kicked up from the road.
Tank materials, then, require hot-chemical resistance. Further, they must endure a broad range of use temperatures and pressures, maintaining integrity as engines and radiators heat up during use and then cool down when vehicles are parked. Because the tanks are exposed on the car’s underside, they also must resist stone impingement lest they crack and leak coolant, which is toxic to humans, animals and the environment. Moreover, the complex shapes of tanks tend to be unique for each platform based on available packaging space, so injection molding is the preferred forming process. Because additional caps or rings/circular disks are often welded onto the tanks after molding to seal off specific areas, good weld strength is critical. Given the limited space in vehicle front ends, mechanical integrity and the ability to hold dimensional tolerances are key material characteristics. To top it off, DuPont Automotive had a growing family of renewable products sourced from agricultural feedstocks, including wheat, corn, soy, sugarcane and castor beans, but at the beginning of this project its bio-based PA 6/10 was primarily used to make filaments for many types of brushes. It had no history in high-temperature, chemically aggressive environments.
Given these engineering challenges, the punishing underhood environment, the fact that a radiator is crucial to safe engine operation and keeping in mind the less-than-stellar reputation of many previous bio-plastics, the team focused a lot of attention on durability. This was defined by DENSO’s specific internal targets for impact strength, chemical resistance, creep resistance and long-term heat-age performance. An additional challenge was the perception that bio-plastics cost more but perform worse than the conventional polymers they replace — a lose/lose proposition, especially after the global economic crash of 2008, when automakers were sitting on product and drastically curtailing production. Although this was DENSO’s first foray into biopolymers, the tank engineering team was convinced that if the high-cost/low-durability paradigm could be reversed, then the impact of this application would be great and could spur greater use of more environmentally friendly materials.
Using the company’s previous radiator end tanks in both PA 6/6 and the blend of PA 6/6 with 6/12 as benchmarks, the team worked to formulate the new bio-based PA 6/10 polymer for long-term durability and manufacturability. Because DENSO also would mold the parts, the company wanted to ensure that the new formulation could be fabricated in the same way as the formulations used in the other end tanks. Throughout the development process, formulations were subjected to DENSO’s standard testing procedure for evaluating new materials, with particular emphasis given to protocols that included ISO-527-1,2 and ISO-179-1,2, as well as a careful assessment of quality and durability.
According to Shinya Goto, manager of the Non-Metallic Materials Department in DENSO’s Materials Engineering R&D Div., the new bio-PA 6/10 formulation bests the conventional PA 6/6 and the 6/6 with 6/12 in elongation and fatigue-resistance: “We were able to obtain twice the durability with the new bio-PA 6/10 material vs. what we had previously achieved with our conventional blend of PA 6/6 with 6/12. Moreover, cost has also been reduced, so our development goals were all successfully met.” Reportedly, PA 6/10 is fully recyclable, just like ordinary thermoplastics.
Having broken the high-cost/low-performance paradigm, and with a viable, proven material in hand, DENSO approached its customer, Toyota, and proposed the first commercial application, which was released on a new version of the Camry. The DENSO/DuPont team’s careful work in development paid off in the smooth changeover to the new radiator end tanks.
Toward a greener future
So successful was this first bio-based radiator end tank that DENSO and DuPont continued their collaboration. Earlier this year a new-generation radiator, featuring bio-based end tanks made of the same material, was announced on Toyota’s luxury Lexus GS. This time around, thanks to a clever but proprietary new design, the radiator system is reportedly 40 percent smaller and lighter, yet it is 10 percent more efficient than DENSO’s previous units.
According to Akio Shikamura, senior executive director of DENSO’s Thermal Systems Business Group, “The radiator’s reduced size translates into greater design flexibility for installation in the engine compartment. This also helps make the vehicle safer in the event of a frontal collision because the smaller size allows for a larger impact or crash zone.”
Since then, DENSO has been evaluating the bio-based PA 6/10 for other uses and is looking at other bio-based materials to use in its operations. The big-picture potential for bio-composites and other bio-based polymer applications was perhaps best summed up by Goto: “The society independent of oil would be ideal,” he says. “The dream would be to build a car of the future using plant-based materials.” |
Related Content
The state of recycled carbon fiber
As the need for carbon fiber rises, can recycling fill the gap?
Read MoreInfinite Composites: Type V tanks for space, hydrogen, automotive and more
After a decade of proving its linerless, weight-saving composite tanks with NASA and more than 30 aerospace companies, this CryoSphere pioneer is scaling for growth in commercial space and sustainable transportation on Earth.
Read MoreProtecting EV motors more efficiently
Motors for electric vehicles are expected to benefit from Trelleborg’s thermoplastic composite rotor sleeve design, which advances materials and processes to produce a lightweight, energy-efficient component.
Read MoreMaterials & Processes: Fibers for composites
The structural properties of composite materials are derived primarily from the fiber reinforcement. Fiber types, their manufacture, their uses and the end-market applications in which they find most use are described.
Read MoreRead Next
Engineering Insights: BMC Creates New Dimensions in Underhood Apps
Specially formulated material enables production of high-tolerance automotive engine air intake component.
Read MoreFrom the CW Archives: The tale of the thermoplastic cryotank
In 2006, guest columnist Bob Hartunian related the story of his efforts two decades prior, while at McDonnell Douglas, to develop a thermoplastic composite crytank for hydrogen storage. He learned a lot of lessons.
Read MoreComposites end markets: Energy (2024)
Composites are used widely in oil/gas, wind and other renewable energy applications. Despite market challenges, growth potential and innovation for composites continue.
Read More





















.jpg;maxWidth=300;quality=90)