Benzoxazines for OOA tooling
Can infusible formulations of this newer resin system produce aerospace-capable tools?
Autoclave cure produces composite parts of the highest quality and lowest void content. All other things being equal, it’s still the best insurance against part rejection. But today, all things aren’t equal. The world in which part performance is everything and cost is no object is passing away, even in previously “cost-plus” defense and space-exploration sectors. Demand for faster production, particularly in the part-per-minute world of automotive composites, has exposed autoclave cure, with its lengthy temperature ramps, to the charge of “production bottleneck.” Part consolidation and elimination of labor-intensive assembly steps — strategies made possible by the nature of composites and which justify their higher cost — have progressively increased the size of composite parts. Corresponding increases in autoclave volume have caused the autoclave’s purchase price and operational costs to skyrocket out of reach for all but the biggest, most well-funded fabricators (read more in “Out-of-autoclave prepregs: Hype or revoltuion?" under "Editor's Picks,” at top right). Further, regardless of part size, the autoclave can make pre-production development cost-prohibitive and can discourage prototyping of tooling and parts, especially in prospective and/or one-off or few-of-a-kind programs.
These realities have motivated the development of out-of-autoclave (OOA) epoxy prepreg (epoxy prepreg is the material form of choice for most autoclaved parts) and bismaleimide (BMI)-based alternatives that can be oven-cured. OOA epoxies and BMIs have seen significant development in tooling applications, but a relative newcomer, benzoxazine, continues to gain ground since structural composites-capable formulations were introduced in 2008 (see “BMI and benzoxazine battle for future OOA aerocomposites," under "Editor's Picks”).
Hot/wet performer
Benzoxazine boasts continuous hot/wet service temperatures, a key parameter in tooling applications, as high as or greater than epoxy’s maximum hot/wet service temperature and also poses less risk of runaway exotherm that could contribute to tool distortion. Further, it can match BMI’s performance in tools (e,g., <1% springback) and has the practical advantage of a significantly longer outlife — 6 months at room temperature.
These factors were the motivation for recent tests on nonproduction tooling manufactured from carbon fiber and benzoxazine resin via vacuum infusion. Toolmaker Leading Edge Aerospace (LEA, Wichita, KS, US) partnered on the R&D project with an aerospace parts manufacturer to examine the viability of the infusion process as a substitute for prototype and production tooling typically made from prepreg cured in an autoclave.
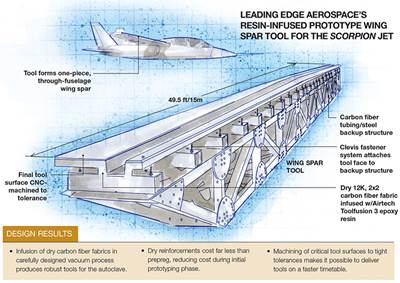
LEA had previously produced tooling via vacuum infusion for other customers. A notable instance involved autoclave-capable infused tools for the wingskin, spars and other composite components that were incorporated into the Scorpion, a tactical fighter jet concept independently developed by Textron Airland LLC (Wichita, KS, US; see the two Scorpion-related articles under “Editor's Picks”). The Scorpion tools combined a dry layup of 12K carbon fiber 2x2 twill fabric and an infusion-optimized epoxy resin, followed by a 12-hour cure under vacuum and a postcure. The use of benzoxazine for this new project enabled LEA and its partner to evaluate the performance of a tooling material that promised increased toughness and better high-temperature performance, compared with epoxy systems.
A female master model was CNC-machined from a 254-mm by 1,016-mm by 1,219-mm block of monolithic graphite. Next, approximately 12 plies of 2x2, 12K carbon fiber fabric were layed up on the master, bagged and vacuum infused with Beta Benzoxazine resin, supplied by US-based Airtech International (Huntington Beach, CA, US). The material has a Tg of 250°C, low water absorption and very low volumetric shrinkage on cure.
Rod Brown, composites manager at LEA, says the resin, as supplied in pails, is highly viscous, and required pre-heating to enable transfer into a feed tank equipped with a thermocouple-controlled temperature controller and lines for infusion and. There, the resin was stirred and gradually ramped up to the infusion temperature of 110°C. As the resin neared the infusion temperature, the tank was fitted with a lid and a vacuum was applied to de-gas the resin.
In-oven infusion
When the resin was fully de-gassed, the tank was placed in the oven, in which the master had been pre-heated to the resin temperature. Resin feed lines were run from the tank to ports in the bagged layup. Operators then had a pot-life window of about an hour in which to infuse the layup. After infusion was complete, the oven temperature was ramped up to the cure temperature of 185°C and held for 4 hours. After cure, an egg crate-type backup structure was attached and the mold was removed from the master. Then the mold and backup structure, together, were postcured at 218°C for about 8 hours.
“The biggest challenge, working with this resin, is how to manage the material, lines and mold at the various temperatures,” says Brown, noting that at 110°C, “you can still walk in the oven, briefly.” That’s not possible at the cure and postcure temperatures. “So everything has to be staged and ready to go,” he cautions.
After postcure, the mold was CNC-machined with a 5-axis unit supplied by Anderson America Corp. (Pineville, NC, US) and then sanded, operations that were followed by application of filler and releases supplied by Chem-Trend (Howell, MI, US).
The finished tool, approximately 76.2 cm by 76.2 cm, was actually a reproduction of a corner section of a tool for a much larger production part. The benzoxazine-infused mold was used to make test parts, each comprising 30 plies of Cytec 934 carbon/epoxy prepreg supplied by Cytec Aerospace Materials (Woodland Park, NJ, US). The parts were autoclave-cured at 177°C and 6.21 bar.
“The tool performed great and maintained vacuum integrity through 15 test cycles,” reports Brown. He adds that the decision to use benzoxazine rather than BMI to infuse the tool wasn’t a strategic decision. “Both materials are very similar in terms of performance and cost,” he notes. “We just tested benzoxazine because that’s what Airtech offers. Our ultimate objective was to find some alternatives to prepreg and the autoclave.”
Large aerocomposites?
That said, Brown sees the results of the project as promising. Although LEA and its aerospace partner have no plans, currently, to fund additional testing, he sees benzoxazine as an OOA tooling alternative for aircraft stiffeners, stringers and wing spars. “The real need for something like this is large tooling because that’s where you enter into problems with an autoclave,” he contends.
But he believes that eventual commercialization of OOA technology involving benzoxazine will hinge, in part, on how benefits and costs weigh out: The upside of using benzoxazine rather than epoxy to infuse tooling is that it produces finished tools that are more durable and can operate at higher temperatures. The downside is that it costs more and is trickier to infuse and cure at the necessarily higher temperatures required to ensure that it will have a viscosity low enough to fully wet out the reinforcement. Although the upside might have enough appeal among aerospace manufacturers to encourage its adoption, more trials and testing will be necessary to pave the path if it is to progress to commercial reality.
Related Content
Materials & Processes: Fabrication methods
There are numerous methods for fabricating composite components. Selection of a method for a particular part, therefore, will depend on the materials, the part design and end-use or application. Here's a guide to selection.
Read MoreA new era for ceramic matrix composites
CMC is expanding, with new fiber production in Europe, faster processes and higher temperature materials enabling applications for industry, hypersonics and New Space.
Read MoreCarbon fiber in pressure vessels for hydrogen
The emerging H2 economy drives tank development for aircraft, ships and gas transport.
Read MoreLarge-format 3D printing enables toolless, rapid production for AUVs
Dive Technologies started by 3D printing prototypes of its composite autonomous underwater vehicles, but AM became the solution for customizable, toolless production.
Read MoreRead Next
BMI and benzoxazine battle for future OOA aerocomposites
Offering weight, cost and process advantages, these “hot zone” resins are moving down the thermometer and into out-of-autoclave structural applications and autoclavable tooling now dominated by epoxies.
Read MoreOut-of-autoclave prepregs: Hype or revolution?
Oven-cured, vacuum-bagged prepregs show promise in production primary structures.
Read MoreResin infusion produces autoclave-capable tools for Scorpion jet
Leading Edge Aerospace designs and builds cost-effective prototype tools for maverick military aircraft concept.
Read More