Resin matrices: Thermosets
Unsaturated polyester resins are the most widely used in commercial, mass-production applications, thanks to their ease of handling, good balance of mechanical, electrical and chemical properties and relatively low cost. Typically coupled with glass fiber reinforcements, polyesters adapt well to a range of fabrication
Unsaturated polyester resins are the most widely used in commercial, mass-production applications, thanks to their ease of handling, good balance of mechanical, electrical and chemical properties and relatively low cost. Typically coupled with glass fiber reinforcements, polyesters adapt well to a range of fabrication processes and are most commonly used in open-mold sprayup, compression molding, resin transfer molding (RTM) and casting. Polyesters provide the primary resin matrix used in bulk molding compounds (BMC) and sheet molding compounds (SMC), materials used in compression molding (see "High-volume molding methods,"p. 26).
The properties of polyester formulations can be modified to meet specific performance criteria, based on the selection of glycol and acid elements and reactive monomers (most commonly, styrene). Polyester resins are often differentiated in terms of their base ingredients. Orthopolyesters, for example, build on orthophthalic acid. Isopolyester resins have isophthalic acid as their essential ingredient, and exhibit superior chemical and thermal resistance compared to orthopolyesters. Terephthalic resins incorporate terephthalic acids and have been formulated for improved toughness, compared to traditional isopolyesters.
Specially formulated, unreinforced polyester resins, known as gel coats, improve the impact and abrasion resistance and the surface appearance of the final product. These are applied to a mold surface and gelled before layup of the composite. In the tub and shower market, for instance, gel-coated fiberglass products have been dominant, and their use continues to grow, despite strong competition from glass/acrylic units made with polymethyl methacrylate (PMMA).
Vinyl ester resins offer a bridge between lower-cost, rapid-curing and easily processed polyesters and higher-performance epoxy resins (described below). Vinyl esters shrink less during cure and outperform polyesters in chemically corrosive environments (e.g., chemical tanks) and in structural laminates requiring a high degree of moisture resistance (such as boat hulls and decks), which accounts, in part, for their higher price.
Cure of these thermosets is exothermic; as they crosslink, they release heat. Fabricators can control the cure profile in terms of shelf life, pot life (the time prior to cure), gel time, cure temperature and viscosity through careful formulation of the catalyst package, which may include inhibitors, promoters and accelerators.
For advanced composite matrices, the most common thermosets are epoxies, phenolics, cyanate esters (CEs), bismaleimides (BMIs) and polyimides.
Epoxy resins contribute strength, durability and chemical resistance to a composite. They offer high performance at elevated temperatures, with hot/wet service temperatures up to 121°C/250°F. Epoxies come in liquid, solid and semisolid forms and typically cure by reaction with amines or anhydrides. Most commercial epoxies have a chemical structure based on diglycidyl ether of bisphenol A or creosol, and/or phenolic novolacs. Many aerospace applications use amine-cured, multifunctional epoxies that require cure at elevated temperatures. Toughening agents, e.g., thermoplastics and reactive rubber compounds, can be added to counteract brittleness.
Phenolic resins are based on a combination of an aromatic alcohol and an aldehyde, such as phenol, combined with formaldehyde. They find application in flame-resistant aircraft interior panels and in commercial markets that require low-cost, flame-resistant and low-smoke products. Excellent char yield and ablative (heat-absorbing) characteristics have made phenolics long-time favorites for ablative and rocket nozzle applications. They have also proven successful in nonaerospace applications, notably in components for offshore oil and gas platforms, and in mass transit and electronics applications. Phenolics, however, release water vapor and formaldehyde during cure, which can produce voids in the composite. As a result, their mechanical properties are somewhat lower than those of epoxies and most other high-performance resins. Molds must be designed with adequate venting or a "breathe"step to allow the water vapor to escape.
Cyanate esters (CEs) are versatile matrices that provide excellent strength and toughness, allow very low moisture absorption and possess superior electrical properties, compared to other polymer matrices, although at a higher cost. CEs feature hot/wet service temperatures to 149°C/300°F and are usually toughened with thermoplastics or spherical rubber particles. They process similarly to epoxies, but their curing process is simpler, thanks to CE's viscosity profile and nominal volatiles. Applications range from radomes, antennae, missiles and ablatives to microelectronics and microwave products.
Among the more exotic of resins, bismaleimide (BMI) and polyimide (close relatives, chemically) are used in high-temperature applications on aircraft and missiles (e.g., for jet engine nacelle components). BMIs offer hot/wet service temperatures (to 232°C/450°F), while some polyimides can be used to 371°C/700°F for short periods of time. Volatiles and moisture emitted during cure make polyimides more difficult to work with than epoxies or CEs; special formulation and processing techniques have been developed to reduce or eliminate voids and delaminations. Both BMIs and polyimides exhibit higher moisture absorption and lower toughness values than CEs or epoxies, but significant progress has been made in recent years to create tougher formulations.
Polybutadiene resins offer good electrical properties and chemical resistance and have been used successfully as alternatives to epoxy in E-glass/epoxy composites typically used to mold thin-walled, glass-reinforced radomes.
Benzoxazines, a subclass of phenolic resins, are formed by reacting a phenol with an aldehyde and an aromatic amine. While the chemistry has been known since the 1940s, Huntsman Advanced Materials (Woodland, Texas) has more recently developed a family of benzoxalines for advanced composites and electronics applications. Another, lesser known resin class is phthalonitriles, originally developed by the U.S. Naval Research Laboratory for very high temperature applications. Commercialized by Eikos Inc. (Franklin, Mass.), phthalonitriles have service temperatures approaching 371°C/700°F, and have been selected for high-temperature engine parts as well as submarine vessels.
Related Content
Composite resins price change report
CW’s running summary of resin price change announcements from major material suppliers that serve the composites manufacturing industry.
Read MoreMaterials & Processes: Fibers for composites
The structural properties of composite materials are derived primarily from the fiber reinforcement. Fiber types, their manufacture, their uses and the end-market applications in which they find most use are described.
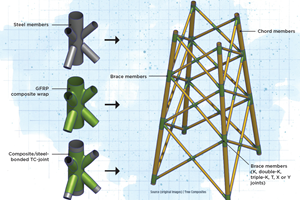
Read MoreNovel composite technology replaces welded joints in tubular structures
The Tree Composites TC-joint replaces traditional welding in jacket foundations for offshore wind turbine generator applications, advancing the world’s quest for fast, sustainable energy deployment.
Read MorePEEK vs. PEKK vs. PAEK and continuous compression molding
Suppliers of thermoplastics and carbon fiber chime in regarding PEEK vs. PEKK, and now PAEK, as well as in-situ consolidation — the supply chain for thermoplastic tape composites continues to evolve.
Read MoreRead Next
From the CW Archives: The tale of the thermoplastic cryotank
In 2006, guest columnist Bob Hartunian related the story of his efforts two decades prior, while at McDonnell Douglas, to develop a thermoplastic composite crytank for hydrogen storage. He learned a lot of lessons.
Read MoreCW’s 2024 Top Shops survey offers new approach to benchmarking
Respondents that complete the survey by April 30, 2024, have the chance to be recognized as an honoree.
Read MoreComposites end markets: Energy (2024)
Composites are used widely in oil/gas, wind and other renewable energy applications. Despite market challenges, growth potential and innovation for composites continue.
Read More