Composites in exoskeletons
Wearable robotics need strength, stiffness and lightweight and are forecast at a CAGR of 40-50%. Will composites play a role or be left behind?

|
|
|
| SOURCE: Data from ExoskeletonReport.com. | SOURCE: Bloomberg and Ekso Bionics. | SOURCE: Data from ABI Research. |
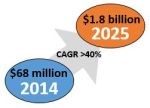
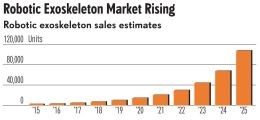
According to a report by ABI Research (Oyster Bay, NY, US), the robotic exoskeleton market is forecast to reach $1.8 billion in 2025, up from $68 million in 2014, with a CAGR of 40%. (Other reports predict a CAGR of almost 50%). Quoting ABI’s report, a Dec 2015 RoboticsTrends.com article notes that lower body exoskeletons used as rehabilitation tools or to improve quality of life are the current market leaders, but commercial systems that augment human capabilities will show the strongest growth moving forward. The latter’s goal is to reduce injuries and improve productivity for worker tasks including heavy lifting, extended standing, squatting, bending or walking. Applications range from construction and agriculture, to transportation and healthcare (e.g. nurses lifting patients). Demand for these wearable robots is predicted to be on par with industrial robots now used widely in manufacturing.
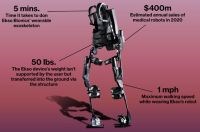
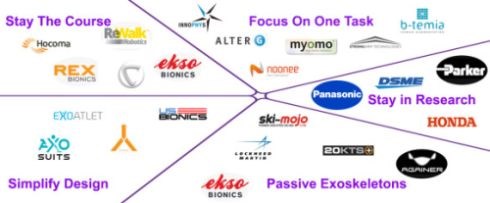
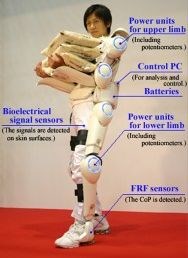
ExoskeletonReport.com lists at least 40 companies working on hard exoskeletons, both powered and unpowered, and more looking into soft exosuits. Carbon fiber composites were used early on by exoskeleton pioneer, Ekso Bionics (Richmond, CA, US) and are also touted for lightweight in Hyundai’s HMEX and Cyberdyne’s HAL for Labor Support. However, most exoskeleton products use very little composites, if any, relying mainly on metals and plastics.
The INDEGO exoskeleton by Parker Hannifin (Cleveland, OH, US) has been approved by the US Food and Drug Administration for use with spinal cord injury patients. “We use carbon fiber in the foot braces,” says director of global communications Aidan Gormley, “but the rest is made from metal and a mix of ABS and polycarbonate plastic.”
INDEGO exoskeletons will initially be supplied to rehabilitation centers, who will manage their use with patients. “The current focus is not lifetime mobility restoration for these patients," says Gormley, "but instead to achieve significant health benefits and efficiencies during rehabilitation by replacing the current robotic treadmill training, which requires numerous physical therapists/attendants, while the INDEGO only requires one.”
|
|
|
|
| SOURCE: Ark Investment Management. | SOURCE: Investor’s Business Daily and ABI Research. |
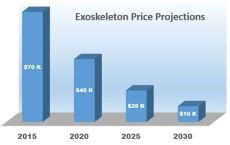
In the same price range as many exoskeleton products, INDEGO costs roughly $80,000. Gormley says personal lifelong use is a definite goal, but the cost is a factor and work is still needed to document its health benefits and usage outcomes in order to obtain health insurance reimbursement. Parker Hannifin has just started a 4-year, multi-center study funded by the U.S. Department of Defense and the Congressionally Directed Medical Research Programs to do just that.

SOURCE: ReWalk.
Ekso Bionics has also received FDA approval, for its Ekso GT product, to be used with stroke and spinal cord injury (SCI) patients, while the US Dept. of Veterans Affairs (VA) has issued a national coverage policy for the use and purchase of ReWalk Robotics Inc. (Marlborough, MA, US) robotic wearable exoskeletons for qualifying veterans with SCI. Veterans will be evaluated for use of the device, which provides powered hip and knee motion, enabling patients to stand upright and walk independently. Training will begin on an outpatient basis and proceed to home or community settings, followed by consideration to procure a personal device for long-term use.
Though both ReWalk and Parker Hannifin are focused primarily on rehabilitation products, Parker is also pursuing human augmentation products via its investment in and partnership with Freedom Innovations (Irvine, CA, US).

|
|

|
| ActiveLink prototype.SOURCE: ibtimes.co.uk. | Cyberdyne’s HAL non-medical exoskeleton. SOURCE: Extremetech.com. | FORTIS exoskeleton. SOURCE: Lockheed Martin |
Human Augmentation and “The Ironman Suit”
The U.S. military has been funding development of an “Iron Man” suit for years. In James Cameron’s movie Avatar, the Mitsubishi MK-6 Amplified Mobility Platform (or “AMP suit”) is described as a descendant of the first military exoskeletons used in the mid-21st century. Though he underestimated the pace of development, Cameron’s science fiction accurately foresaw military and heavy industry participation.

|

|
| The Assist Suit AWN-03 was developed for disaster response and clean-up. SOURCE: ExoskeletonReport.com, Mitsubishi Heavy Industries and JAPC. | Cyberdyne’s HAL for Labor Support is being tested by Daiwa. SOURCE: Cyberdyne. |
Tokyo-based Mitsubishi Heavy Industries and Japan Atomic Power Company have produced the Power Assist Suit for nuclear disasters. Working with Panasonic subsidiary ActiveLink (Nara, Japan), which is 20% owned by Mitsui (Tokyo, Japan), the Assist Suit AWN-03 began selling last September. ActiveLink has also widely showcased its Dual Arm Power Amp and Power Loader products. With arms that extend to 2m and the ability to lift almost 300 kg, early Power prototypes do indeed appear to be the ancestor of the fictional AMP suit in Avatar.
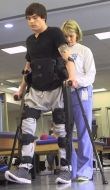
New exoskeleton products are also being tested by Seoul, Korea-based Daewoo Shipbuilding and Marine Engineering and Hyundai Motor Co., as well as Japanese tractor manufacturer Kubota (Osaka) and Japan’s largest homebuilder Daiwa House Industry Co. (Osaka). Daiwa is actually road testing Cyberdyne’s Hybrid Assistive Limb Cybernetic Suit, or HAL. One driver for Japan's heavy investment into exoskeletons is its aging population. Exoskeletons can prolong useful working life and improve productivity as workers age.
On the defense side, Lockheed Martin Exoskeleton Technologies (Orlando, FL, US) first licensed Ekso Bionics’ Human Universal Load Carrier (HULC) and has now unveiled its 3rd generation industrial Human Augmentation System (iHAS) product FORTIS, which it originally developed for US Navy shipbuilders.

|

|
|
Dubbed the “Iron Man Suit”, the US Army’s developmental Tactical Assault Light Operator Suit (TALOS) is aimed to better protect soldiers on the front lines, while DARPA's Warrior Web seeks an under-uniform suit to prevent musculoskeletal injuries and reduce fatigue . SOURCE: (left) TechTimes.com and US Army, (right) Gajitz.com and DARPA. |
|
Ekso Bionics, meanwhile, has won phase I and II contracts from the US Army to develop its next-gen exoskeleton, offically named the Tactical Assault Light Operator Suit (TALOS), but oft referred to as the "Ironman Suit". Slated to have an operational prototype by August 2018, the suit is aimed to better protect soldiers on the front lines.
The Defense Advanced Research Projects Agency (DARPA, Arlington, VA, US), is taking a different approach via its Warrior Web program. It seeks to develop a lightweight, flexible suit that can be worn under a uniform, augmenting and supporting a soldier's muscles to prevent one of the most common reasons for discharge: musculoskeletal injuries. The suit's goals include the ability to detect imminent muscular injuries and react to provide more support, as well as enabling soldiers to run a 4-minute mile (with gear) without fatigue.
Future for Exoskeleton Materials
According to a 2015 article in WIRED magazine, the unpowered Ekso Works Industrial Exoskeleton takes great advantage of carbon fiber: “The trick is the carbon fiber harness and metal-tube frame running down a user’s legs. It translates the weight of whatever’s on the end of the arm down through the suit and into the ground.” However, where early descriptions of Ekso's medical rehabilitation exokeletons touted carbon fiber and aluminum with equal billing, they are now described as mainly aluminum. Powered, they weigh roughly 23 kg including battery.
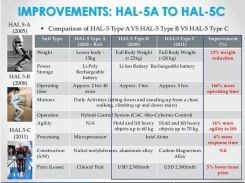
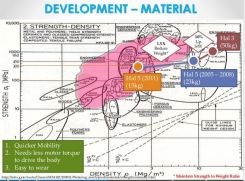
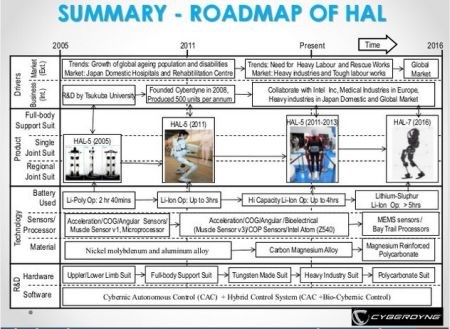
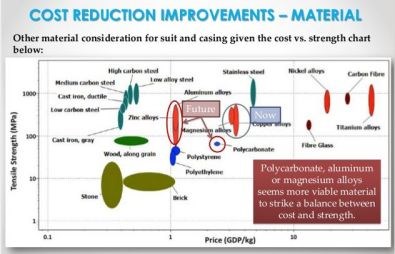
Metal also seems favored by Cyberdyne as they continue to develop their HAL products, with carbon and glass fiber shown as slightly higher in performance, but too high in cost. The slides above and below are from a Cyberdyne presentation "Opportunities in Robotic Exoskeletons," published in 2013 on SlideShare as “Robotic Exoskeletons: Becoming Economically Feasible”. They show that the company has used both metals and composites to reduce weight as it developed its HAL exoskeleton products. The company's latest direction, however, appears to be magnesium-reinforced polycarbonate for a globally marketed product. What’s interesting, is that the tensile strength vs. price chart the Japanese used in this Cyberdyne presentation was actually generated at the University of Cambridge (Cambridge, UK) using software from Granta Design Ltd. (Cambridge, UK). The chart next to it (see above, click to enlarge) is also from the University of Cambridge and shows basically the same results, but use £/kg for cost instead of GDP/kg.
|
|
|
|
Cyberdyne has used developments in both metals and composites to reduce weight in past iterations of its HAL exoskeletons. SOURCE: Cyberdyne presentation on SlideShare: "Robotic Exoskeletons: Becoming Economically Feasible".
In a feature article to be published in the June issue of CW, the world’s largest prosthetics manufacturer, Otto Bock (Duderstadt, Germany), discusses its use of CFRP. It has also licensed two of Ekso Bionics’ patents. Maximilian Segl, head of composite development at Otto Bock, sees a need for improvement in the fiber-to-resin interface of composites to enable thinner and lighter weight parts. Össur (Reykjavik, Iceland), an orthopedic device producer and leader in mind-controlled bionic prosthetics, also uses CFRP, and, according to R&D engineer Christophe Lecomte, sees potential for composites to offer not only lighter and stronger devices, but also prosthetics that could harvest energy while patients walk or self-heal when damaged. Össur is also looking at reducing waste, recycling/recycled materials and improved sustainability.
Will composites have a future in exoskeletons? I think the odds will be substantially higher if the composites industry engages manufacturers. The companies developing and building these devices won’t understand how the latest developments in resins, fibers, processing and recycling could provide solutions if they don’t know these developments exist. We at CW report on so many things that could have real traction here, from recyclable liquid-molded parts via Elium acrylic resin or Recyclamine epoxy hardener, to carbon fiber sizing optimized for thermoplastics, to recycled carbon fiber molding compounds and all kinds of processing developments. As always, we want to support the composites industry and welcome your suggestions and comments. Be sure to read the June issue.
Related Content
Natural fiber composites: Growing to fit sustainability needs
Led by global and industry-wide sustainability goals, commercial interest in flax and hemp fiber-reinforced composites grows into higher-performance, higher-volume applications.
Read MoreWhy aren't composites synonymous with infrastructure?
The U.S. seems poised to invest heavily in infrastructure. Can the composites industry rise to the occasion?
Read MoreThe making of carbon fiber
A look at the process by which precursor becomes carbon fiber through a careful (and mostly proprietary) manipulation of temperature and tension.
Read MoreASCEND program update: Designing next-gen, high-rate auto and aerospace composites
GKN Aerospace, McLaren Automotive and U.K.-based partners share goals and progress aiming at high-rate, Industry 4.0-enabled, sustainable materials and processes.
Read MoreRead Next
From the CW Archives: The tale of the thermoplastic cryotank
In 2006, guest columnist Bob Hartunian related the story of his efforts two decades prior, while at McDonnell Douglas, to develop a thermoplastic composite crytank for hydrogen storage. He learned a lot of lessons.
Read MoreComposites end markets: Energy (2024)
Composites are used widely in oil/gas, wind and other renewable energy applications. Despite market challenges, growth potential and innovation for composites continue.
Read MoreCW’s 2024 Top Shops survey offers new approach to benchmarking
Respondents that complete the survey by April 30, 2024, have the chance to be recognized as an honoree.
Read More



















.jpg;maxWidth=300;quality=90)










